 The "7" is added four times since there are four fluorine atoms with 7 valence electrons each. Then add 4 + 7 + 7 + 7 + 7 to get a total of 32 valence electrons. For instance, if you are trying to find the total number for valence electrons in CF 4, then you will need to first find out that carbon (C) has four valence electrons and fluorine (F) has seven valence electrons. To do so, add up all the valence electrons for each atom in the molecule of that compound. In the periodic table, elements with analogous valence electron configurations usually occur within the same group. Recall that all elements found within the same column on the periodic table have the same number of valence electrons. Once you have determined the number of valence electrons for the atoms of each element in the compound, you are ready to determine the total number of valence electrons in the molecule. Electrons in the outermost orbitals, called valence electrons, are responsible for most of the chemical behavior of elements. A pattern-based 'charge shortcut' does, indeed, exist, in the form of a trend that spans the main group or 'A-Block' columns on the periodic table. Answer: It is not the valence electrons themselves, but the number of valence electrons that determines the chemical properties of an element. Similar logic can be applied to all other groups to obtain the results shown in the graphic below. Group 13 elements have an s 2p 1 electron configuration and three valenece electrons. The Periodic Table was designed with this feature in mind.  
All Group 2 elements have an s 2 electron configuration and two valenece electrons. For this reason, elements with the same number of valence electrons tend to have similar chemical properties, since they tend to gain, lose, or share valence electrons in the same way. 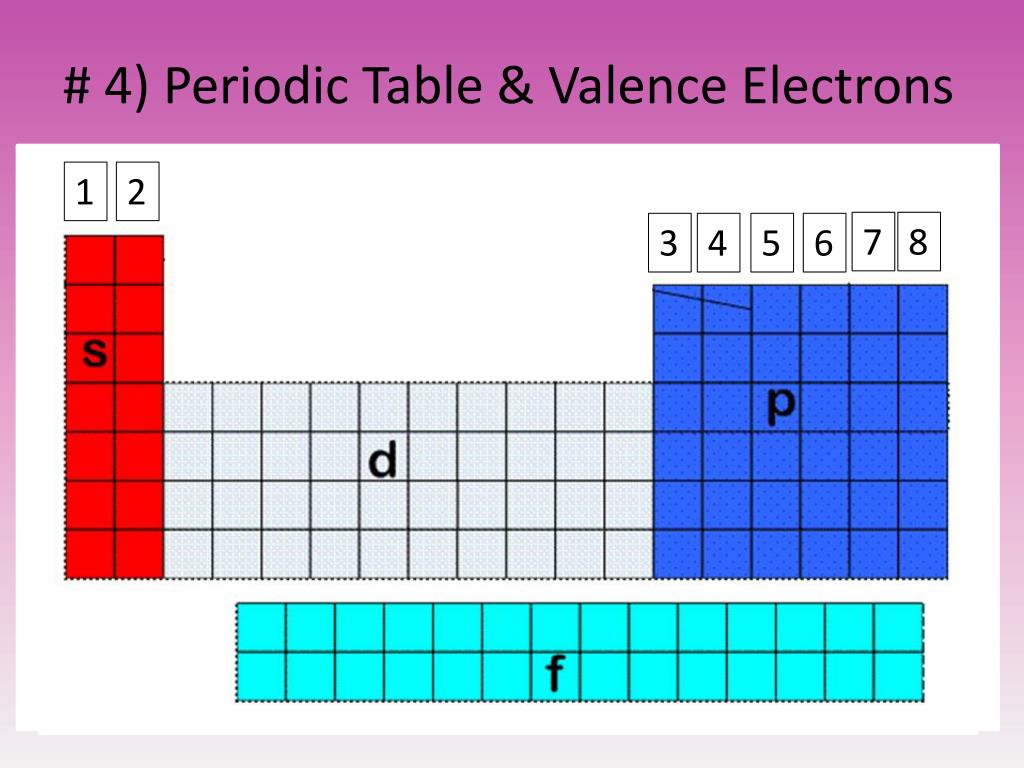
The number of valence electrons in one atom of each element is easily determined based on its position in the periodic table. As shown in the graphic below, all Group 1 elements have an s 1 electron configuration and thus have one valence electron. Valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation of chemical bonds. Since the number of valence electrons is a periodic property, the value can be easily determined by locating the element in the periodic table. The number of dots equals the number of valence electrons in the atom. How does one Determine the # of Valence Electrons?įirst you need to be able to determine the number of valence electrons present in each element of the compound. A Lewis electron dot diagram (or electron dot diagram, or a Lewis diagram, or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
